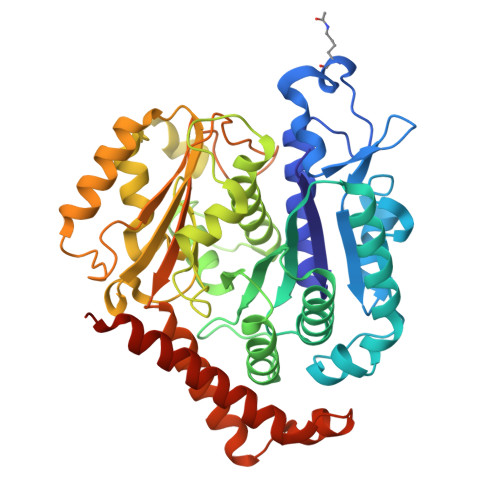
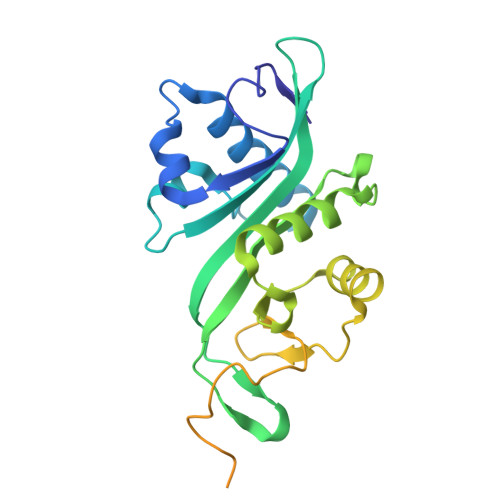
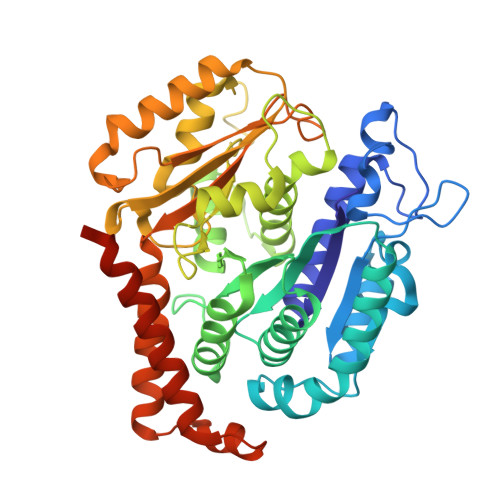
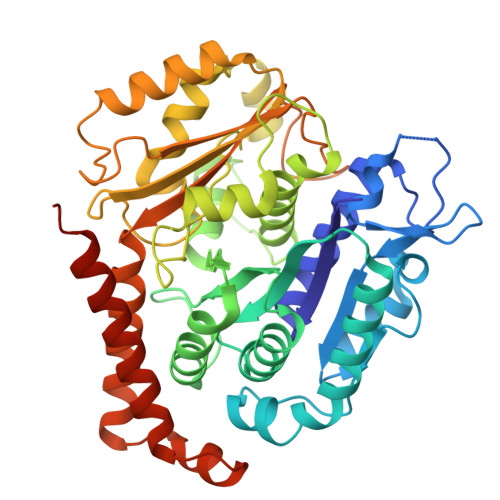
Tubulin acetyltransferases access and modify the microtubule luminal K40 residue through anchors in taxane-binding pockets.
Luo, J., Lam, W.H., Yu, D., Chao, V.C., Zopfi, M.N., Khoo, C.J., Zhao, C., Yan, S., Liu, Z., Li, X.D., Zheng, C., Zhai, Y., Ti, S.C.(2024) Nat Struct Mol Biol
- PubMed: 39496813
- DOI: https://doi.org/10.1038/s41594-024-01406-3
- Primary Citation of Related Structures:
8Y9F, 8YAJ, 8YAL, 8YAR - PubMed Abstract:
Acetylation at α-tubulin K40 is the sole post-translational modification preferred to occur inside the lumen of hollow cylindrical microtubules. However, how tubulin acetyltransferases access the luminal K40 in micrometer-long microtubules remains unknown. Here, we use cryo-electron microscopy and single-molecule reconstitution assays to reveal the enzymatic mechanism for tubulin acetyltransferases to modify K40 in the lumen. One tubulin acetyltransferase spans across the luminal lattice, with the catalytic core docking onto two α-tubulins and the enzyme's C-terminal domain occupying the taxane-binding pockets of two β-tubulins. The luminal accessibility and enzyme processivity of tubulin acetyltransferases are inhibited by paclitaxel, a microtubule-stabilizing chemotherapeutic agent. Characterizations using recombinant tubulins mimicking preacetylated and postacetylated K40 show the crosstalk between microtubule acetylation states and the cofactor acetyl-CoA in enzyme turnover. Our findings provide crucial insights into the conserved multivalent interactions involving α- and β-tubulins to acetylate the confined microtubule lumen.
Organizational Affiliation:
School of Biomedical Sciences, Faculty of Medicine, The University of Hong Kong, Hong Kong SAR, China.